The original data in these publications are compiled in the Data Sheets immediately following this Critical Evaluation. For convenience, further discussion of this system will be in two parts.
1. THE SOLUBILITY OF METHYLCYCLOHEXANE (1) IN WATER (2)
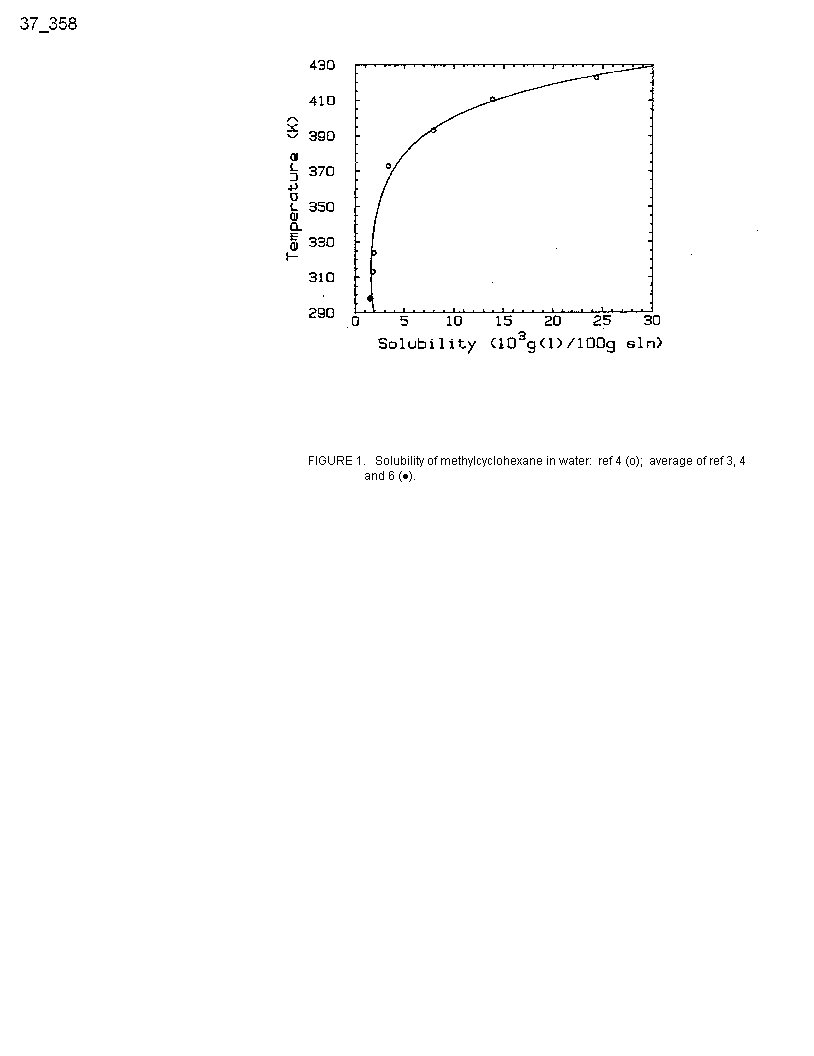
At 298 K the various solubility values (ref 3, 4, 6) are in good agreement (Table 2). The datum of Krzyzanowska and Szeliga (ref 5) has been excluded from consideration because it does not appear to be independent of that of Price (ref 4).
At other temperatures the values of Guseva and Parnov (ref 1), probably obtained in sealed tubes at unspecified pressures, are almost an order of magnitude larger than those of Price (ref 4) and are thus rejected. All other data are listed in Table 2 and plotted in Figure 1.
Roof (ref 7) has reported critical end point at 572.2 K and 3.47 MPa.
2. THE SOLUBILITY OF WATER (2) IN METHYLCYCLOHEXANE (1)
The solubility of water in methylcyclohexane has been reported only by Englin et al. (ref 2) and so no critical evaluation can be made. The interested user is referred to the relevant Data Sheet for solubility values. However, it should be noted that the data of Englin et al. are generally reliable at lower temperatures but are high when T > 300 K.
Figure
References:
1
Englin, B. A.; Plate, A. F.; Tugolukov, V. M. Pryanishnikova, M. A.; Chem. Technol. Fuels Oils, 1965, 1(9), 722-726.
2
McAuliffe, C.; J. Phys. Chem. 1966, 70, 1267-1275.
3
Krzyzanowska, T.; Szeliga, J.; Nafta (Katowice) 1978, 12, 413-7.
4
Rudakov, E. S.; Lutsyk, A. I.; Zh. Fiz. Khim. 1979, 53, 1298-1300.
5
Guseva, A. N.; Parnov, E. I.; Vestn. Mosk. Univ. Khim. 1964, 19, 77-8.
6
Price, L. C.; Am. Assoc. Petrol. Geol. Bull. 1976, 60, 213-44.
7
Roof, J. G.; J. Chem. Eng. Data 1970, 15, 301-3.
